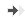Newsletter # 02

Animal and Cellular models
The only approved treatment for ALS is riluzole, which is believed to reduce damage to motor neurons by decreasing the release of glutamate. Clinical trials with ALS patients show that riluzole prolongs survival by several months, mainly in those with difficulty swallowing. The drug also extends the time before a patient needs ventilation support. Riluzole does not reverse the damage already done to motor neurons.
-
Drug Screening :Two screening tests are primarily used for evaluating the therapeutic potential of novel compounds in the treatment of ALS.
-
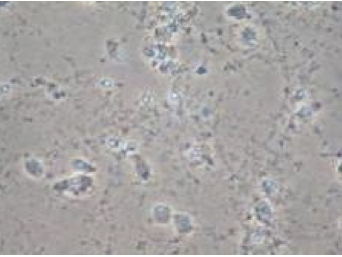
In Vitro Survival assay :A number of findings support the role of glutamate excitotoxicity in the ALS pathophysiology. In this in vitro survival test 13-day-old cortical neuron cultures are injured by an acute intoxication with glutamate or NMDA. Compounds are evaluated for neuroprotective effects by adding them to the culture medium either before (protective) or after (restorative) the application of glutamate or NMDA. Neuronal cell death is determined by measuring LDH activity in the media at 24h after glutamate/NMDA exposure.
-
Cortical neurons
13-day old culture of cortical neuron on control condition. -
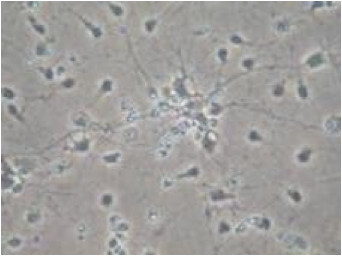
Cortical neurons
13-day old culture of cortical neuron injured by glutamate (10min, 75µM). -
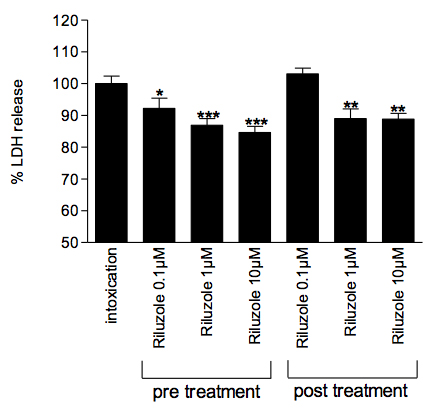
Effect of Riluzole on neuronal death induced by glutamate
-
In vivo SOD1 mouse model of familial ALS :Transgenic mice overexpressing the mutant human form of SOD1 is widely recognized to rapidly develop progressive motor neuron disease that strongly resembles to ALS. At about 2.5 - 3 months of age SOD1 mice demonstrate progressively muscle weakness along with nerve dysfunction indicated by a decrease in the amplitude and an increase in latency of compound muscle action potential (CMAP). By 4 months of age, death by paralysis and respiratory failure appears. In the clinic Riluzole shows a modest extension of survival, but no effect on motor function, and a similar profile is observed in SOD1 mice following chronic treatment with riluzole. For drug screening, compounds are given chronically (typically starting at day 60 of age) and their effects in the SOD1 mice are evaluated by measuring: survival or survival and nerve function (electrophysiology measures) or survival, nerve function and motor performance (behavioural measures).
-
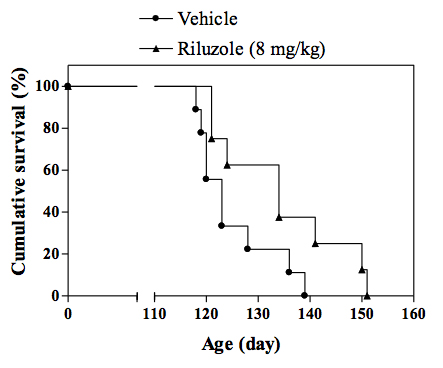
Cumulative survival curve of transgenic SOD1 mice. -
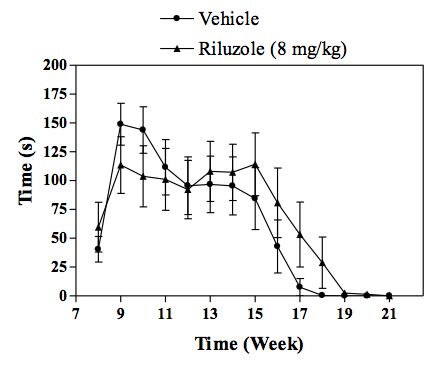
Motor performance of transgenic mice as assessed by the rotarod test.
-
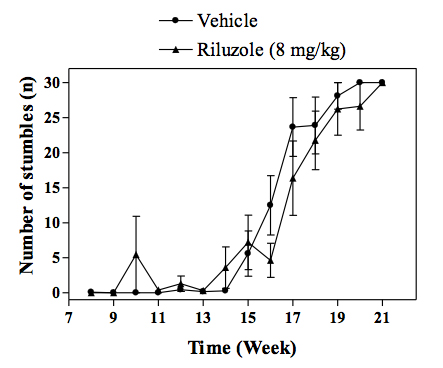
Motor performance of transgenic mice as assessed the grid test. -
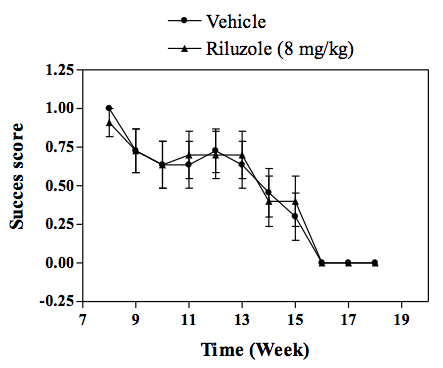
Motor performance of transgenic mice as assessed by the string test.
-
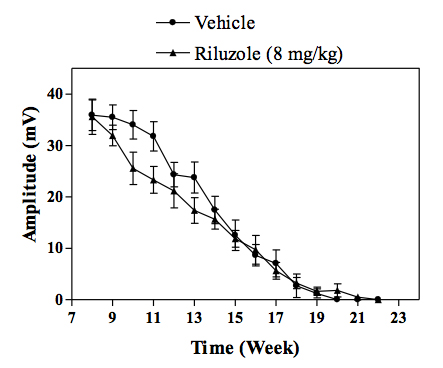
Change in the amplitude of CMAP in transgenic SOD1 mice. -
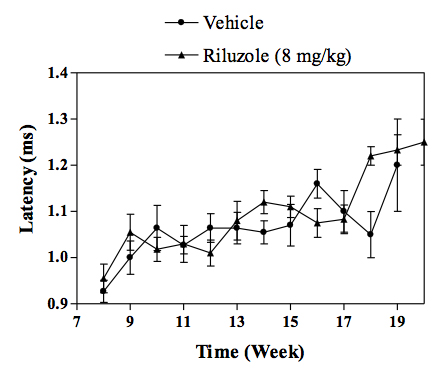
Change in the latency of CMAP in transgenic SOD1 mice.
References :
-
ANDERSEN PM, NILSON P, ALA-HURULA V, KERANEN, ML, TARVAINEN I, HALTIA T, NILSSON L, BINZER M, FORSGREN L, MARKLUND S (1995) Amyotrophic lateral sclerosis associated with homozygosity for an Asp90Ala mutation in Cu,Zn superoxide dismutase. Nature Genet. 10 : 61-66..
BECKMAN JS, CARSON M, SMITH CD, KOPPENOL WH (1993) ALS, SOD and peroxynitrite. Nature 364 : 584.
BROWN RH (1995) Superoxide dismutase in familial amyotrophic sclerosis : models for gain of function. Curr. Opin. Neurobiol. 5 : 841-846.
CHOI D.W., KOH J. and PETERS S. (1988). Pharmacology of glutamate neurotoxicity in cortical cell cultures: attenuation by NMDA antagonists. J. Neurosci., 8, 185-196.
DENG HX, HENTATI A, TAINER JA, IQBAL Z, CAYABYAB A, HUNG WY, GETZOFF ED, HU P, HERZFELDT B, ROOS RP, et al. (1993) Amyotrophic lateral sclerosis and structural defects in Cu,Zn superoxide dismutase. Science 261 : 1047-1051.
GURNEY ME, PU H, CHIU AY, DAL CANTO MC, POLCHOW CY, ALEXANDER DD, CALIENDO J, HENTATI A, KWON YW, DENG HX, et al. (1994) Motor neuron degeneration in mice that express a human Cu,Zn superoxide dismutase mutation. Science 264 : 1772-1775.
-
GURNEY M, CUTTING FB, ZHAI P, DOBLE A, TAYLOR C, ANDRUS PK, HALL ED (1996) Benefit of vitamin E riluzole, and gabapentin in a transgenic model of familial amyotrophic lateral sclerosis. Ann. Neurol. 39 : 147-157.
GURNEY ME (1997) The use of transgenic mouse model of amyotrophic lateral sclerosis in preclinical drug studies. J. Neurol. Sci. 152 : S67-73.
MOHAJERI MH, FIGLEWICZ DA, BOHN MC (1998) Selective loss of alpha motoneurons innervating the medial gastrocnemius muscle in a model of amyotrophic lateral sclerosis. Exp. Neurol. 150 : 329-336.
ROSEN DR, SIDDIQUE T, PATTERSON D, FIGLEWICZ DA, et al. (1993). Mutations in Cu/Zn superoxide dismutase gene are associated with familial amyotrophic lateral sclerosis. Nature 362 : 59-62.
ROTHSTEIN JD, VAN KAMMEN M, LEVEY AI, MARTIN LJ, KUNCL RW (1995) Selective loss of glial glutamate transporter GLT-1 in amyotrophic lateral sclerosis. Ann. Neurol. 38 : 73-84.
YANG G, CHAN PH, CHE J, CARLSON E, CHEN SF, WEINSTEIN P, EPSTEIN CJ, KAMII H (1994) Human copper-zinc superoxide dismutase transgenic mice are highly resistant to perfusion injury after focal cerebral ischemia. Stroke 25 : 165-170.
Get in touch


 PREVIOUS
PREVIOUS