Newsletter # 62

Animal models
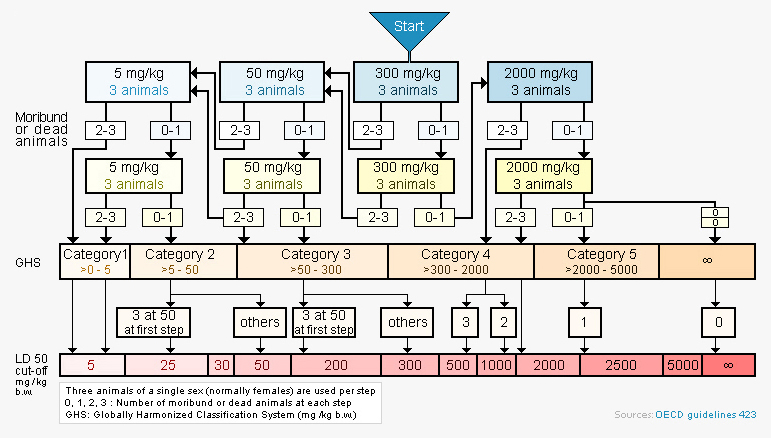
with a starting dose of 300 mg/kg
-
-
Each step in the protocol uses three animals. The absence or presence of compound-related mortality of animals dosed at one step determines the next step. Lethal exposure ranges are rapidly established and an estimation of an LD50 value can be made when at least two doses result in mortality >0% and <100%. This model facilitates rapid ranking of compounds within a series, or across series, by their toxicity and provides an informed guide to choosing doses for IND-enabling studies.
Get in touch


 PREVIOUS
PREVIOUS