Newsletter # 38
Animal models

Relapsing-remitting MS (RRMS) is the most common disease course at the time of diagnosis.
Approximately 85-90% of individuals with MS are initially diagnosed with RRMS.

-
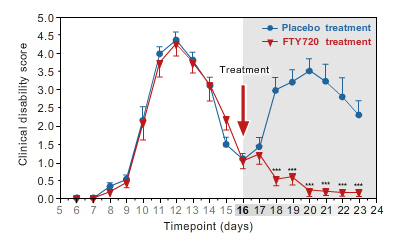
Validated animal model testing for MS with period of remission and relapse, as seen in human MS.
Time course of clinical disability in rats treated with placebo versus rat treated with Fingolimod. Oral treatment with Fingolimod is initiated at remission from the attack
(day 16). -
In this experimental model, the disease relapse is prevented by oral FTY720 (Fingolimod : Gilenya®), a clinically approved drug for MS.
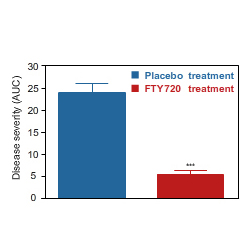
Comparison of disease severity in placebo and Fingolimod-treated rats.
This model also responds to DEXAMATHASONE, another clinically effective drug for the treatment of acute relapse.
Get in touch


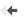 PREVIOUS
PREVIOUS